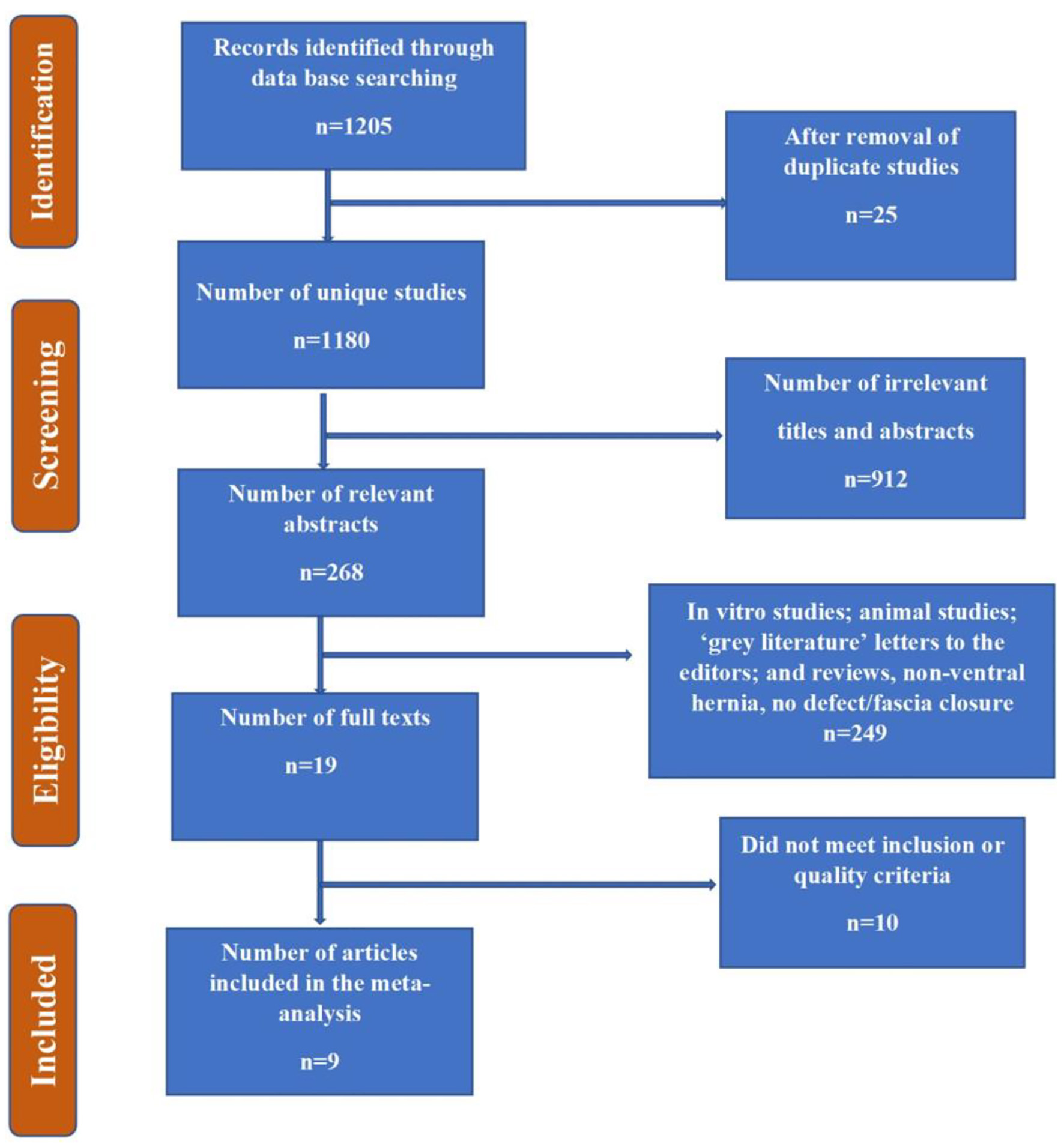
Figure 1. Flow chart summarizing the identification and selection of papers for systematic review.
| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://www.jofem.org |
Review
Volume 12, Number 1, February 2022, pages 10-18
Understanding the Role of Dipeptidyl Peptidase-4 Inhibitors in COVID-19: Findings From a Systematic Review
Figure

Table
| Section and topic | Checklist item | Location where item is reported |
|---|---|---|
| Title | Identify the report as a systematic review. | Abstract, Introduction and Methods |
| Abstract | See the PRISMA 2020 for Abstracts checklist. | Seen and followed this guideline |
| Rationale | Describe the rationale for the review in the context of existing knowledge. | Rationale is described. |
| Objectives | Provide an explicit statement of the objective(s) or question(s) the review addresses. | Objectives are described. |
| Eligibility criteria | Specify the inclusion and exclusion criteria for the review and how studies were grouped for the syntheses. | All required details are in Methods section. |
| Information sources | Specify all databases, registers, websites, organizations, reference lists and other sources searched or consulted to identify studies. Specify the date when each source was last searched or consulted. | Databases are specified in Methods section. |
| Search strategy | Present the full search strategies for all databases, registers and websites, including any filters and limits used. | A full table of search strategy is made and details are in Methods section. |
| Selection process | Specify the methods used to decide whether a study met the inclusion criteria of the review, including how many reviewers screened each record and each report retrieved, whether they worked independently, and if applicable, details of automation tools used in the process. | All required details are in Methods section. |
| Data collection process | Specify the methods used to collect data from reports, including how many reviewers collected data from each report, whether they worked independently, any processes for obtaining or confirming data from study investigators, and if applicable, details of automation tools used in the process. | All required details are in Methods section. |
| Data items | List and define all outcomes for which data were sought. Specify whether all results that were compatible with each outcome domain in each study were sought (e.g., for all measures, time points, analyses), and if not, the methods used to decide which results to collect. | All required details are in Methods section. |
| List and define all other variables for which data were sought (e.g., participant and intervention characteristics, funding sources). Describe any assumptions made about any missing or unclear information. | These are listed in Methods as well as Tables. | |
| Effect measures | Specify for each outcome the effect measure(s) (e.g., risk ratio, mean difference) used in the synthesis or presentation of results. | It is specified in Tables. |
| Synthesis methods | Describe the processes used to decide which studies were eligible for each synthesis (e.g., tabulating the study intervention characteristics and comparing against the planned groups for each synthesis (item #5)). | Flow chart is made with details in Methods. |
| Describe any methods required to prepare the data for presentation or synthesis, such as handling of missing summary statistics, or data conversions. | NA | |
| Describe any methods used to tabulate or visually display results of individual studies and syntheses. | See the flow chart and Tables. | |
| Describe any methods used to synthesize results and provide a rationale for the choice(s). If meta-analysis was performed, describe the model(s), method(s) to identify the presence and extent of statistical heterogeneity, and software package(s) used. | Since it was not a meta-analysis, we performed qualitative review. | |
| Describe any methods used to explore possible causes of heterogeneity among study results (e.g. subgroup analysis, meta-regression). | NA | |
| Describe any sensitivity analyses conducted to assess robustness of the synthesized results. | NA | |
| Reporting bias assessment | Describe any methods used to assess risk of bias due to missing results in a synthesis (arising from reporting biases). | NA |
| Certainty assessment | Describe any methods used to assess certainty (or confidence) in the body of evidence for an outcome. | NA |
| Study selection | Describe the results of the search and selection process, from the number of records identified in the search to the number of studies included in the review, ideally using a flow diagram. | Flow chart and details are in Methods. |
| Cite studies that might appear to meet the inclusion criteria, but which were excluded, and explain why they were excluded. | Flow chart and details are in Methods. | |
| Study characteristics | Cite each included study and present its characteristics. | Description is given in Tables and Results section. |
| Results of individual studies | For all outcomes, present, for each study: (a) summary statistics for each group (where appropriate) and (b) an effect estimate and its precision (e.g. confidence/credible interval), ideally using structured tables or plots. | Description is given in Tables and Results section. |
| Discussion | Provide a general interpretation of the results in the context of other evidence. | Done. |
| Discuss any limitations of the evidence included in the review. | Done. | |
| Discuss any limitations of the review processes used. | Done. | |
| Discuss implications of the results for practice, policy, and future research. | Done. |